|
For example, an aqueous solution of ammonia should be written NH 3(aq), not NH 4 +(aq) + OH -(aq) or as NH 4OH(aq). By the way, these comments also apply to weak bases. The take-a-way is that you need to know which acids are strong and which are weak. Even though some ions are present, the most common procedure is to write weak acids as molecules. Typically, weak acids are one to five percent ionized, meaning that, in solution, they exist mostly as molecules. However, weak acids should be written as molecules, not as ions. Both of these are stong acids and ionize 100%, so I wrote them as ions. 
In previous problems, I used HCl and HNO 3. This is because it is a weak acid and, in solution, ionizes only to a small extent (somewhere in the 1% to 5% amount, it varies due to the specific acid and its concentration). Notice that the phosphoric acid was written in a molecular way. 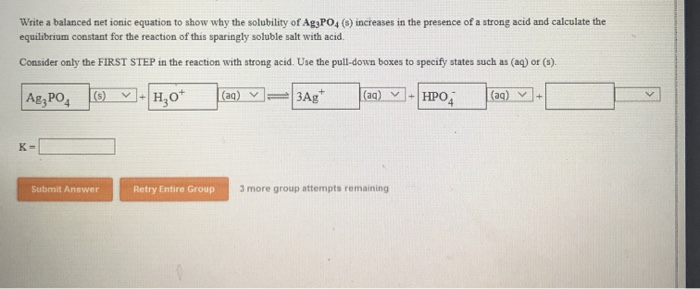
Problem #7: Write the complete ionic and net ionic equations for: In reality, every one molecule of H 2SO 4 that reacts requires 2 formula units of NaOH. Here lies a potential problem when you go to acid base topics: in terms of moles, sulfuric acid and sodium hydroxide react in a one to two molar ratio, NOT a one to one ratio as the net ionic seems to say. Problem #6: Write the complete ionic and net ionic equations for: Problem #5: Write the complete ionic and net ionic equations for: You know that PbSO 4 precipitates from using a solubility chart. Solutions of lead(II) chloride and potassium sulfate react to form a precipitate of lead(II) sulfate and aqueous potassium chloride Problem #4: Write the complete molecular, complete ionic and net ionic equations for: HCl(g) are gas molecules floating freely in space. Be careful, because you could see HCl(g) in an example. When a substance is a gas, it is always molecular, never ionic. Yes, you do need to recognize that H 2S is a gas and, as such, is a molecular substance. NaCl is the quintessential ionic substance. HCl is a strong acid and ionizes 100% in solution. Recognize sodium sulfide as a soluble, ionic substance. Solutions of sodium sulfide and hydrochloric acid react to form sodium chloride and hydrogen sulfide 
Problem #3: Write the complete molecular, complete ionic and net ionic equations for: Problem #2: Write the following equation as a complete ionic equation and a net ionic equation:īr 2(ℓ) + 2NaI(aq) -> I 2(s) + 2NaBr(aq)īr 2(l) + 2Na +(aq) + 2I¯(aq) -> I 2(s) + 2Na +(aq) + 2Br¯(aq) You have to recognize AlBr 3 as ionic and H 2 as molecular.ĢAl(s) + 6H +(aq) -> 2Al 3+(aq) + 3H 2↑ You have to know that HBr is a strong acid and, as such, will ionize 100% in solution. Problem #1: Write the following equation as a complete ionic equation and a net ionic equation:ĢAl(s) + 6HBr(aq) -> 2AlBr 3(aq) + 3H 2↑ĢAl(s) + 6H +(aq) + 6Br¯(aq) -> 2Al 3+(aq) + 6Br¯(aq) + 3H 2↑ 
NH 4NO 3 + Pb(ClO 4) 2 -> Pb(NO 3) 2 + NH 4ClO 4Įvery compound is soluble and every compound ionizes 100%. Here are two NR that I just decided to put here: ChemTeam: Complete Molecular, Complete Ionic and Net Ionic: Ten Problems Complete Molecular, Complete Ionic and Net Ionic: Ten Problems Fifteen Problems Twenty-Five Problems Net ionic equation tutorial All NR Equations & Reaction Types menu
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
